Article Text
Abstract
Objective Our study aims to present a summary of the clinicopathological characteristics of patients affected by the coronavirus disease 2019 (COVID-19) that can be used as a reference for further research and clinical decisions.
Design Studies were included in the meta-analysis if they had cohort, case–control or case series designs and provided sufficient details on clinical symptoms, laboratory outcomes and asymptomatic patients.
Setting PubMed, Embase, Chinese Biomedical Literature Database, Wanfang, China Science and Technology Journal Database and China National Knowledge Infrastructure databases were electronically searched to identify related studies published between 1 January 2020 and 16 March 2020. Three reviewers independently examined the literature, extracted relevant data and assessed the risk of publication bias before including the studies in the meta-analysis.
Participants The confirmed cases of COVID-19.
Results A total of 55 unique retrospective studies involving 8697 patients with COVID-19 were identified. Meta-analysis showed that a higher proportion of infected patients were male (53.3%), and the two major symptoms observed were fever (78.4%) and cough (58.3%). Other common symptoms included fatigue (34%), myalgia (21.9%), expectoration (23.7%), anorexia (22.9%), chest tightness (22.9%) and dyspnoea (20.6%). Minor symptoms included nausea and vomiting (6.6%), diarrhoea (8.2%), headache (11.3%), pharyngalgia (11.6%), shivering (15.2%) and rhinorrhea (7.3%). About 5.4% of the patients were asymptomatic. Most patients showed normal leucocyte counts (64.7%) and elevated C reactive protein levels (65.9%). Lymphopaenia was observed in about 47.6% of the infected patients, along with abnormal levels of myocardial enzymes (49.4%) and liver function (26.4%). Other findings included leucopenia (23.5%), elevated D-dimer (20.4%), elevated erythrocyte sedimentation rate (20.4%), leucocytosis (9.9%), elevated procalcitonin (16.7%) and abnormal renal function (10.9%).
Conclusions The most commonly experienced symptoms of patients with COVID-19 were fever and cough. Myalgia, anorexia, chest tightness and dyspnoea were found in some patients. A relatively small percentage of patients were asymptomatic and could act as carriers of the disease. Most patients showed normal leucocyte counts, elevated levels of C reactive protein and lymphopaenia, confirming the viral origin of the disease.
- communicable disease control
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction
In the spring of 2020, the coronavirus disease 2019 (COVID-19) pandemic has spread to more than 200 countries around the world.1 2 As of 27 March 2020, the total number of confirmed cases has exceeded 500 000.3 This pandemic has become a serious threat to global health and continues to challenge healthcare systems worldwide. It was determined to be caused by a novel coronavirus, the severe acute respiratory syndrome coronavirus 2.4 Therefore, it is critical to understand and identify the key clinical and laboratory characteristics of patients with COVID-19 in order to help in early detection and isolation of infected individuals, as well as minimise the spread of the disease.5
Although a number of studies have attempted to explore this subject, most of them were single-centre studies that were conducted in a specific hospital or region. Due to differences in study design and small samples, the clinical symptoms, laboratory findings and other key outcomes of these studies are complicated and unclear.6–8 For example, two recent systematic reviews9 10 of studies of patients with COVID-19 indicated a high incidence of fever (>88%) and cough (>68%), but only one10 reported symptoms of myalgia or fatigue (35.8%). Both reviews meta-analysed small samples pooled from 10 studies.
Therefore, the present meta-analysis was performed to provide the most extensive, up-to-date description so far of clinicopathological characteristics of patients with COVID-19 and to provide a reference for clinical decisions and future research.
Materials and methods
Search strategy and study eligibility
This meta-analysis was carried out based on the guidelines of the Preferred Reporting Items for Meta-Analyses of Observational Studies in Epidemiology Statement.11 We systematically examined the studies on clinicopathological characteristics of patients with COVID-19 indexed in the PubMed, Embase, Chinese Biomedical Literature, Wanfang, China Science and Technology Journal Database and China National Knowledge Infrastructure databases between 1 January 2020 and 16 March 2020. All references cited in these studies were also analysed manually to ensure that eligible papers were not overlooked. If multiple studies analysed the same patient population, we included only the one with more detailed information or the one published more recently. No language restrictions were incorporated during the literature search, and only literature published online was included. The following keywords were used, both separately and in combination, as part of the search strategy in each database: ‘Corona virus’, ‘Coronavirus’, ‘2019-nCoV’, ‘COVID-19’ or ‘SARS-CoV-2’ (box 1).
Search strings used for the PubMed database
# 1 Corona virus [Title/Abstract]
# 2 Coronavirus [Title/Abstract]
# 3 2019-nCoV [Title/Abstract]
# 4 COVID-19 [Title/Abstract]
# 5 SARS-CoV-2 [Title/Abstract]
# 6 # 1 OR # 2 OR # 3 OR # 4 OR # 5
Studies were included in the meta-analysis if they had cohort, case–control or case series designs and provided sufficient details on clinical symptoms, laboratory outcomes and asymptomatic patients. Only studies of more than 40 patients were included.
Data extraction and quality assessment
The literature selected was independently assessed by three reviewers based on the eligibility criteria, and relevant data were extracted. Disagreements were resolved by consensus. The titles and abstracts were first screened to identify the eligible articles, followed by a full-text review to obtain detailed information. When required, the authors were contacted directly to obtain further information and clarifications regarding their study. The following data were extracted from each included study: surname of first author; date of publication; study design; number, age and sex of patients; clinical and laboratory outcomes; and data relevant for assessing publication bias. The quality of observational case series was independently evaluated by the three reviewers based on the British National Institute for Clinical Excellence12 guidelines. This evaluation was conducted based on a set of eight criteria, and studies with a score greater than 4 were considered to be of high quality (total score=8).
Statistical analyses
All statistical analyses were performed using STATA V.12. Original incidence rates r were transformed by the double arcsine method to ensure a normal distribution, and the resulting transformed rate tr was used in single-arm meta-analysis. The heterogeneity between studies was analysed using a χ2 test (p<0.10) and quantified using the I2 statistic. When no statistical heterogeneity was observed, a fixed-effects model was used. Otherwise, potential sources of clinical heterogeneity were identified using subgroup and sensitivity analyses; these sources were eliminated, and the meta-analysis was repeated using a random-effects model. Pooled incidence rates R were back-calculated from transformed rates tr using the R=[sin (tr / 2)].2 A two-tailed p<0.05 was considered statistically significant. Publication bias was evaluated using a funnel plot along with Egger’s regression test and Begg’s test.
Results
Literature screening and assessment
A total of 5576 records were identified from the various databases examined. After a detailed assessment based on the inclusion criteria, 55 unique studies6–8 13–64 involving 8697 patients with COVID-19 were included in the meta-analysis (figure 1).
Flow chart depicting literature screening process.
Characteristics of included studies
All studies included in the meta-analysis were conducted in China and published between 6 February 2020 and 16 March 2020. These retrospective studies examined Chinese patients distributed across 31 provinces. A large proportion of these studies (n=40) were based on data collected from a single centre, with no clear explanation regarding eligibility criteria. Follow-up data were reported for most patients. All studies received quality scores of 5–8, indicating high quality (table 1).
Characteristics of included studies
Meta-analysis results
Gender distribution
Relevant data regarding the clinicopathological characteristics of 8697 patients with COVID-19 was collected.6–8 13–64 Significant heterogeneity was observed across all included studies (I2=93.7%), therefore, a random-effects model was used in the meta-analysis. We found that 53.3% (95%CI 50.3 to 56.4) of the patients were male.
Clinical symptoms
Two major symptoms, including fever (78.4%) and cough (58.3%), were highly prevalent among patients. Fatigue (34%), myalgia (21.9%), expectoration (23.7%), anorexia (22.9%), chest tightness (22.9%) and dyspnoea (20.6%) also occurred frequently. Less frequent symptoms were nausea and vomiting (6.6%), diarrhoea (8.2%), headache (11.3%), pharyngalgia (11.6%), shivering (15.2%) and rhinorrhea (7.3%). Only 5.4% of patients with COVID-19 were found to be asymptomatic (table 2).
Clinical symptoms observed in patients with COVID-19
Pathological characteristics
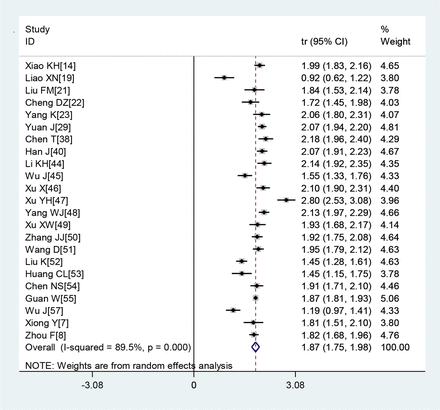
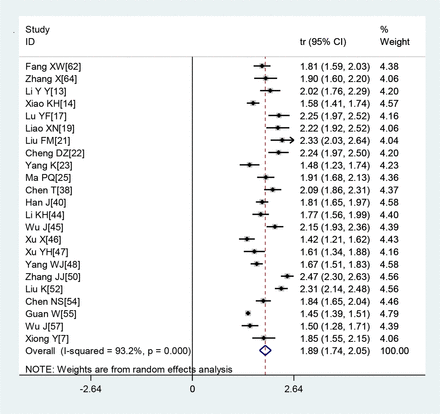
A large proportion of patients had normal leucocyte counts (64.7%) and high levels of C reactive protein (65.9%) (figures 2 and 3). Lymphopaenia was observed in many patients (47.6%), along with elevated levels of myocardial enzymes (49.4%) and abnormal liver function (26.4%). Also observed were leucopenia (23.5%), leucocytosis (9.9%), abnormal renal function (10.9%), elevated levels of D-dimer (20.4%), elevated erythrocyte sedimentation rate (20.4%) and elevated procalcitonin (16.7%) (table 3).
Pathological characteristics of patients with COVID-19
Transformed incidence rate of normal leucocyte count in patients with COVID-19.
Transformed incidence rate of high C reactive protein levels in patients with COVID-19.
Subgroup analysis
Patients were stratified into two groups based on the date of initial diagnosis: group 1 included all patients and group 2 included those diagnosed between December 2019 and 31 January 2020 (table 4). We found that all patients diagnosed before 31 January had higher incidence rates of fever and cough. No significant difference was observed in the heterogeneity between the subgroups and the overall heterogeneity, indicating that the date of initial diagnosis was not the main source of heterogeneity.
Analysis of clinical symptoms observed in patients with COVID-19, stratified by date of initial diagnosis*
Sensitivity analysis
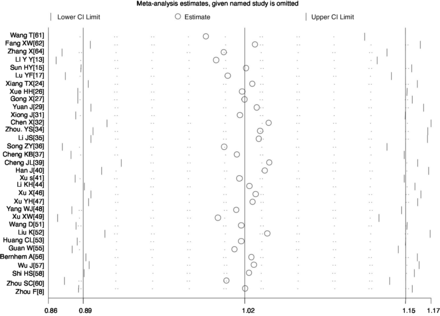
A sensitivity analysis was carried out by excluding one study at a time and reanalysing the entire dataset. We found that the pooled incidence rates did not change substantially, indicating the reliability and stability of our meta-analysis (eg, figure 4).
Sensitivity analysis of the incidence rate of expectoration in patients with COVID-19.
Evaluation of publication bias
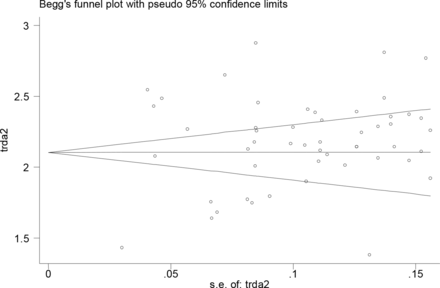
The p values derived using the Egger’s and the Begg’s test for all the clinicopathological characteristics showed no obvious publication bias (table 5). A funnel plot based on the incidence rate of fever showed p values of 0.091 in Egger’s test and 0.703 in Begg’s test (figure 5). These results confirm that there is no publication bias.
Evaluation of publication bias using the Egger’s and the Begg’s test
Evaluation of publication bias using a funnel plot based on the incidence rate of fever.
Discussion
In this meta-analysis, we examined 55 independent studies6–8 13–64 reporting clinicopathological data on 8697 patients with COVID-19 distributed across 31 provinces in China. The studies included in this analysis comprise the latest research available on COVID-19 through 16 March 2020. Our results indicate that there is a slightly higher proportion of male patients (53.3%) and that the main symptoms of this disease are fever (78.4%), cough (58.3%) and fatigue (34%). Compared with previous results,9 10 our findings reveal lower incidence rates of the two major symptoms of this disease, which we found to depend to some extent on whether diagnosis was made before or after 31 January 2020, reflecting with the progress of the epidemic, the number of atypical manifestations has growed gradually. For example, some patients developed gastrointestinal symptoms, such as diarrhoea, nausea and vomiting. These results highlight the importance of also taking into account non-respiratory symptoms of the disease.
Most patients with COVID-19 showed normal leucocyte counts and lymphopqenia. Few patients had leucocytosis and elevated procalcitonin levels, confirming that this disease is transmitted by a virus. Therefore, it is essential for clinicians to use such pathological findings to rule out the presence of bacterial infections. In this study, 49.4% of the patients presented with myocardial enzyme spectrum abnormalities, which manifested as an increase in lactate dehydrogenase levels. Studies have shown that elevated levels of lactate dehydrogenase can be a risk factor for rapid progression from mild to critical COVID-19.65 Therefore, monitoring the function of important organs during treatment is critical, and treatment should be adjusted as needed to preserve and maintain organ function.
Infected people who are asymptomatic can act as a source of infection,66 especially since the estimated median incubation period is 5–6 days (range 0–14 days). An analysis by the Chinese Center for Disease Control and Prevention conducted through 17 February 2020 suggested that the proportion of asymptomatic patients was only around 1%,67 but our results suggest that the proportion is closer to 5%. This increase may reflect the growing experience of hospitals with this novel disease and increasing screening of suspected COVID-19 cases for viral infection, allowing the correct diagnosis of greater proportions of patients showing no or less typical manifestations. Therefore, to control the spread of this disease, general practitioners should carefully monitor individuals with histories of contact in areas where outbreaks have occurred or who had contact with suspected or confirmed cases of COVID-19 within 14 days before onset of symptoms.68 Epidemiological history of patients should be investigated in detail, and asymptomatic infected people in the community should be identified as quickly as possible to control spread of the disease.
A recent study suggests that, considering different scenarios, highly effective contact tracing and case isolation are sufficient to control a new outbreak of COVID-19 within 3 months.69 Therefore, isolation, quarantine, social distancing, and community containment measures should be rapidly implemented in high-risk countries or regions.70 In China, community engagement has been the first line of defence in the battle against the COVID-19 pandemic. General practitioners act as both gatekeepers and health promoters by educating the public and guiding the community in the fight against this disease.71 Monitoring people at designated checkpoints, intercepting transmission routes in a timely manner and preventing local outbreaks are critical to prevent repeat epidemics.72
Although this study rigorously analysed clinical and laboratory data collected from a large sample of patients with COVID-19, we were unable to eliminate the significant heterogeneity observed between studies. For example, the course and the severity of the disease varied across studies. Given that most of the studies included in our meta-analysis were single-centre, retrospective studies, it was difficult for us to control for the effects of several confounding factors, including bias in patient admission and selection, as well as differences in disease severity and course. Further research is required to verify and extend our results for China. Continued surveillance across multiple countries, along with transparent and accurate reporting of patient characteristics and testing policies, will help us gain a better understanding of this global pandemic.73
Conclusion
In summary, Current evidence showed that the most commonly experienced symptoms of patients with COVID-19 were fever and cough. Myalgia, anorexia, chest tightness and dyspnoea were found in some patients. A relatively small percentage of patients were asymptomatic and could act as carriers of the disease. Most patients showed normal leucocyte counts, elevated levels of C reactive protein and lymphopenia, confirming the viral origin of the disease. Due to limited quality and quantity of the included studies, more high-quality prospective studies are required to verify above conclusions.
Footnotes
Correction notice This article has been corrected since it was published Online First. The article type has been modified from 'Original research' to 'Review'.
Contributors PJ, JP and BL collected and analysed the data. HL helped collect the data. JZ acquired the funding. JZ designed the study, and wrote the first draft of the manuscript. CZ and JZ designed and supervised the study, and finalised the manuscript, which all authors read and approved.
Funding This study was supported by grants from the National Natural Science Foundation of China (81960343); the Emergency Science and Technology Brainstorm Project for the Prevention and Control of COVID-19, which is part of the Guangxi Key Research and Development Plan (2020AB39028).
Competing interests None declared.
Patient consent for publication Not required.
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement Data are available on reasonable request.